Drsoccerball
Well-Known Member
- Joined
- May 28, 2014
- Messages
- 3,647
- Gender
- Undisclosed
- HSC
- 2015
Does this sound right ?
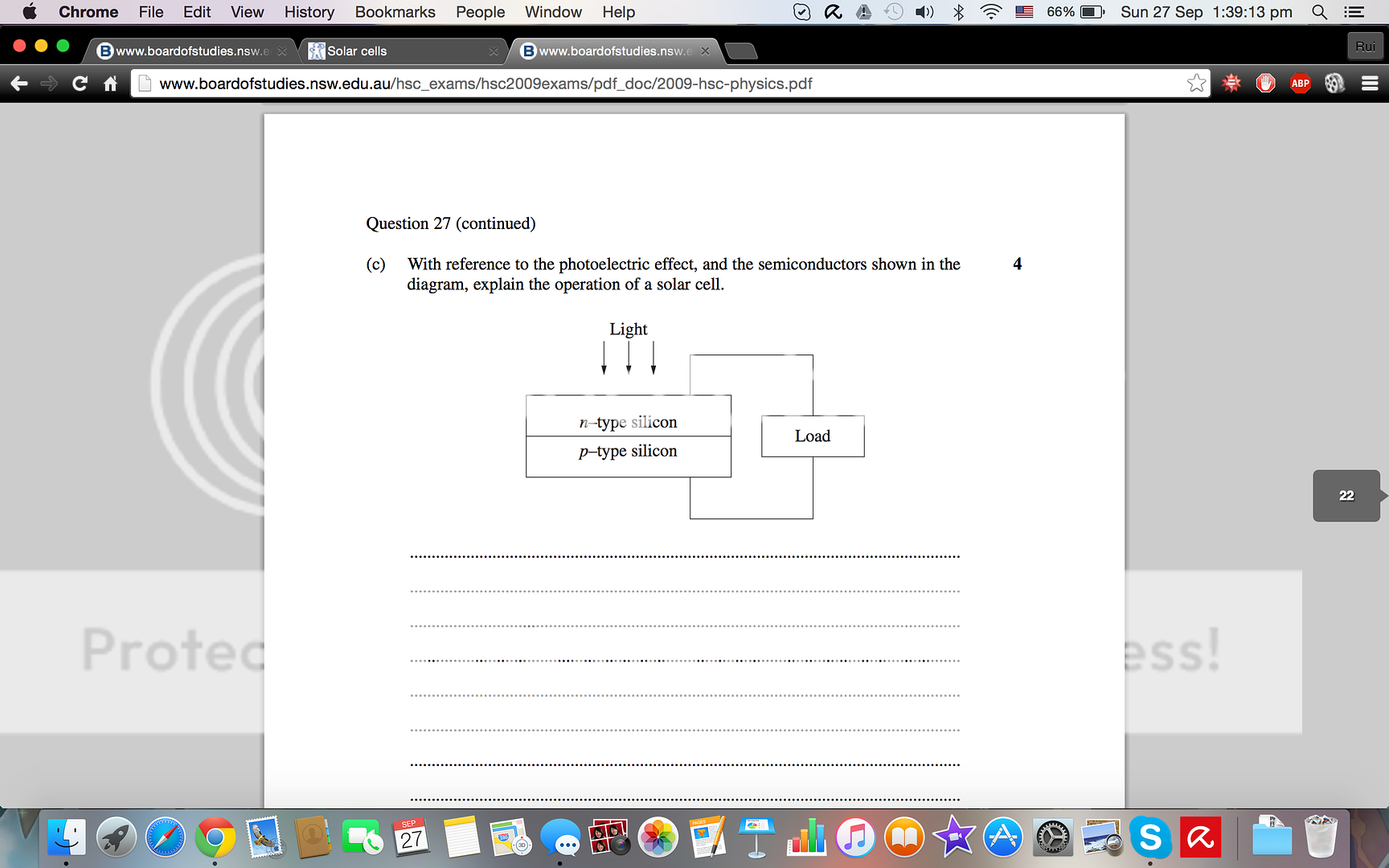
When light hits an n type semi-conductor, if the frequency is higher than the work function electrons will be emitted towards the p-type semi-conductor from the n-type. Light must hit the n-type semiconductor as the electrons are in the n-type thus it requires significantly less energy to circulate around the circuit. The energy provided also has to be high enough to prevent the recombination of electrons.
When light hits an n type semi-conductor, if the frequency is higher than the work function electrons will be emitted towards the p-type semi-conductor from the n-type. Light must hit the n-type semiconductor as the electrons are in the n-type thus it requires significantly less energy to circulate around the circuit. The energy provided also has to be high enough to prevent the recombination of electrons.