In terms of a quick review of these two dot points:
Explain the overall observations about equilibrium in terms of the collision theory
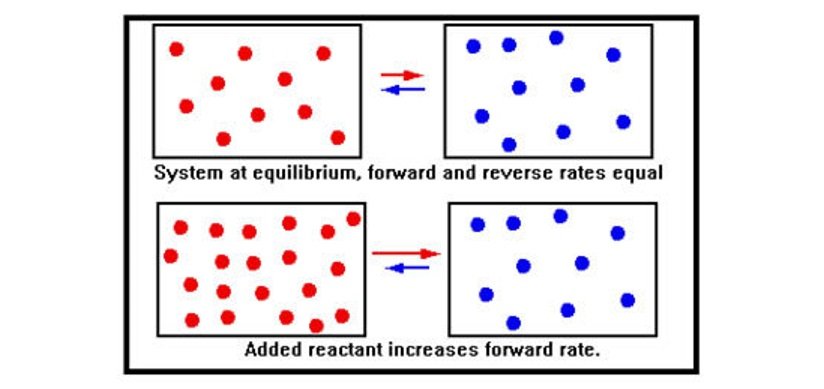
Essentially, chemical equilibrium occurs when opposing reactions proceed at equal rates: The rate at which the products form from the reactants equals the rate at which the reactants form from the products. When additional reactant is added to a system at equilibrium, the immediate effect is to increase the concentration of reactant molecules.
As per the collision theory, this increases the number of collisions per second of reactant molecules and therefore the rate of the forward reaction.
As the concentrations change, the forward rate falls, the reverse rate rises, and eventually the two rates become equal – equilibrium is reestablished with some of the added reactant having been converted to product.
Examine how activation energy and heat of reaction affect the position of equilibrium
Recall the collision theory - the kinetic energy of a gas is directly proportional to temperature. As temperature increases, molecules gain energy and move faster and faster resulting in higher probability that molecules will be moving with the necessary activation energy for a reaction to occur upon collision.
The dot point mentions two concepts, the first being activation energy. This is the minimum energy with which reactants must collide in order for a reaction to occur. The source of the activation energy needed to push reactions forward is typically heat energy from the surroundings.
The second is the heat of reaction, which is the change in the enthalpy of a chemical reaction. In endothermic reactions,
(ΔH>0), heat is absorbed with the reactants. For example:
}+6\:H_2O_{\left(l\right)\:})

}+6\:O_{2\:\left(g\right)})
Consequently, more energy is needed to overcome the forces of attraction between molecules and to separate them from one another (the activation energy) than the energy gained when new bonds are formed.
In exothermic reactions,
(ΔH<0), heat is released with the products. When separated molecules join together, enough energy is released to overcompensate for the energy required to break reactant bonds. In this oxidation reaction:
}+H_2O)

_{2\:\left(s\right)}+\text{heat})
The forward reaction is exothermic because energy is released when
CaO(s) and
H2O(l) combine to form
_2(s))
. The energy to break the bonds of each reactant is lower than the amount of energy released from forming the product, and the net difference is observed as heat on the right side of the equation.
From the above information, it can be inferred that a temperature increase favours an endothermic reaction, and a temperature decrease favours an exothermic reaction.
Since I did not take Chemistry, I sadly can't advise on ways through which you can link those to the Module 7 concepts that you have mentioned. Perhaps someone else could assist you with that more closely.
I hope this helps!