-
Looking for HSC notes and resources? Check out our Notes & Resources page
Make a Difference – Donate to Bored of Studies!
Students helping students, join us in improving Bored of Studies by donating and supporting future students!
please help!!! (titration chemistry) (1 Viewer)
- Thread starter moniyoo
- Start date
nightweaver066
Well-Known Member
- Joined
- Jul 7, 2010
- Messages
- 1,585
- Gender
- Male
- HSC
- 2012
These should keep you busy for a while. I haven't decided on whether they're good or not, but difficulty is relative and i hope this practise is beneficial and will help you for your practical test. 
A standard solution was prepared by dissolving 1.432g of sodium carbonate in water. The solution was made up to 250mL in a volumetric flask. Calculate the concentration of the sodium carbonate solution.The solution was used to determine the concentration of a solution of hydrochloric acid. Four 25.00mL samples of acid were titrated with the sodium carbonate solution. The average titration volume was 22.35mL. Why was it necessary to conduct four titrations? Calculate the concentration of the hydrochloric acid solution.
10. (2 marks)
During a titration, the acid and base are usually mixed in a flask. This flask is rinsed with water only and not with the
solution to be placed in it, explain why this is done.
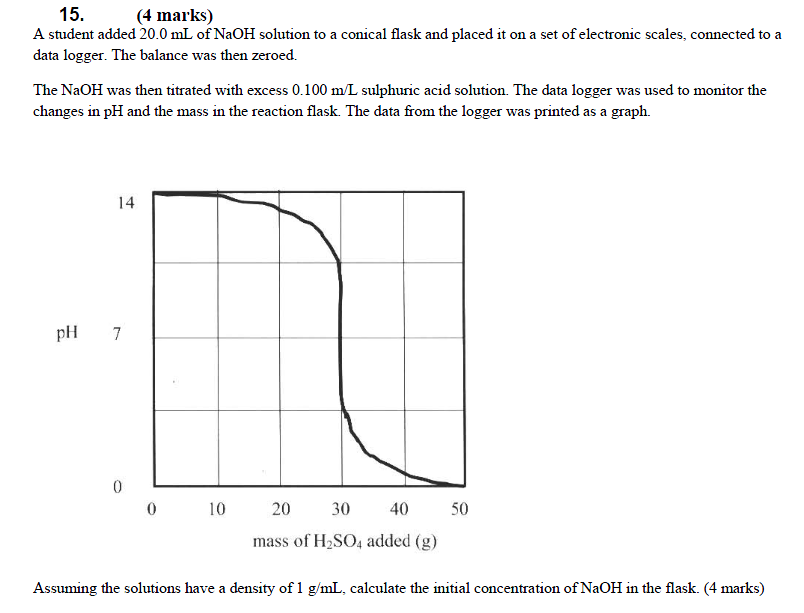
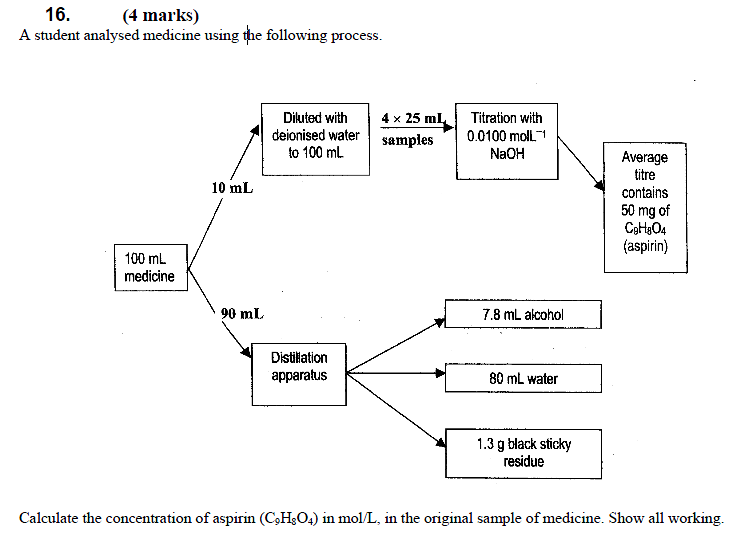
17. (4 marks)
Find the volume of 0.15 mol/L NaOH that will neutralise 25 mL of 0.06 mol/L sulfuric acid.

20. (2 marks)
A student performed a titration using NaOH and orange juice. Explain why phenolphthalein is a more suitable indicator
than methyl orange.


A standard solution was prepared by dissolving 1.432g of sodium carbonate in water. The solution was made up to 250mL in a volumetric flask. Calculate the concentration of the sodium carbonate solution.The solution was used to determine the concentration of a solution of hydrochloric acid. Four 25.00mL samples of acid were titrated with the sodium carbonate solution. The average titration volume was 22.35mL. Why was it necessary to conduct four titrations? Calculate the concentration of the hydrochloric acid solution.
10. (2 marks)
During a titration, the acid and base are usually mixed in a flask. This flask is rinsed with water only and not with the
solution to be placed in it, explain why this is done.


17. (4 marks)
Find the volume of 0.15 mol/L NaOH that will neutralise 25 mL of 0.06 mol/L sulfuric acid.

20. (2 marks)
A student performed a titration using NaOH and orange juice. Explain why phenolphthalein is a more suitable indicator
than methyl orange.


Last edited:
oh thank you thank you !!!Try doing HSC 2010 Q28. Look at the marking guidelines and sample answer if you need help with it...
wow.... they are really good thank you so much!These should keep you busy for a while. I haven't decided on whether they're good or not, but difficulty is relative and i hope this practise is beneficial and will help you for your practical test.
A standard solution was prepared by dissolving 1.432g of sodium carbonate in water. The solution was made up to 250mL in a volumetric flask. Calculate the concentration of the sodium carbonate solution.The solution was used to determine the concentration of a solution of hydrochloric acid. Four 25.00mL samples of acid were titrated with the sodium carbonate solution. The average titration volume was 22.35mL. Why was it necessary to conduct four titrations? Calculate the concentration of the hydrochloric acid solution.
10. (2 marks)
During a titration, the acid and base are usually mixed in a flask. This flask is rinsed with water only and not with the
solution to be placed in it, explain why this is done.


17. (4 marks)
Find the volume of 0.15 mol/L NaOH that will neutralise 25 mL of 0.06 mol/L sulfuric acid.

20. (2 marks)
A student performed a titration using NaOH and orange juice. Explain why phenolphthalein is a more suitable indicator
than methyl orange.


nightweaver066
Well-Known Member
- Joined
- Jul 7, 2010
- Messages
- 1,585
- Gender
- Male
- HSC
- 2012
Oh and the 2011 HSC question on titration is pretty good.
oh okok !thanks!! ummm but what question... may i ask?Oh and the 2011 HSC question on titration is pretty good.
nightweaver066
Well-Known Member
- Joined
- Jul 7, 2010
- Messages
- 1,585
- Gender
- Male
- HSC
- 2012
Q26oh okok !thanks!! ummm but what question... may i ask?
arifmosavi
New Member
- Joined
- Aug 8, 2015
- Messages
- 1
- Gender
- Male
- HSC
- 2016
hi what is the answerThese should keep you busy for a while. I haven't decided on whether they're good or not, but difficulty is relative and i hope this practise is beneficial and will help you for your practical test.
A standard solution was prepared by dissolving 1.432g of sodium carbonate in water. The solution was made up to 250mL in a volumetric flask. Calculate the concentration of the sodium carbonate solution.The solution was used to determine the concentration of a solution of hydrochloric acid. Four 25.00mL samples of acid were titrated with the sodium carbonate solution. The average titration volume was 22.35mL. Why was it necessary to conduct four titrations? Calculate the concentration of the hydrochloric acid solution.
10. (2 marks)
During a titration, the acid and base are usually mixed in a flask. This flask is rinsed with water only and not with the
solution to be placed in it, explain why this is done.


17. (4 marks)
Find the volume of 0.15 mol/L NaOH that will neutralise 25 mL of 0.06 mol/L sulfuric acid.

20. (2 marks)
A student performed a titration using NaOH and orange juice. Explain why phenolphthalein is a more suitable indicator
than methyl orange.



